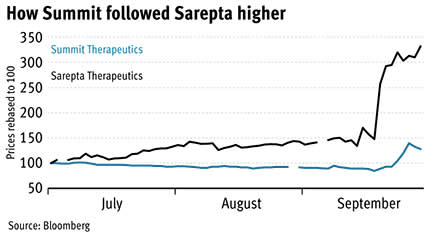
That’s good news for Aim-traded Summit Therapeutics (SUMM), whose DMD drug Ezutromid is in the second phase of clinical trials. On Monday Ezutromid was granted fast-track designation by the FDA and a day later was recognised as a rare paediatric disease drug, which will allow it to move through clinical trials quickly.
 Search
Search
Join our community of smart investors